














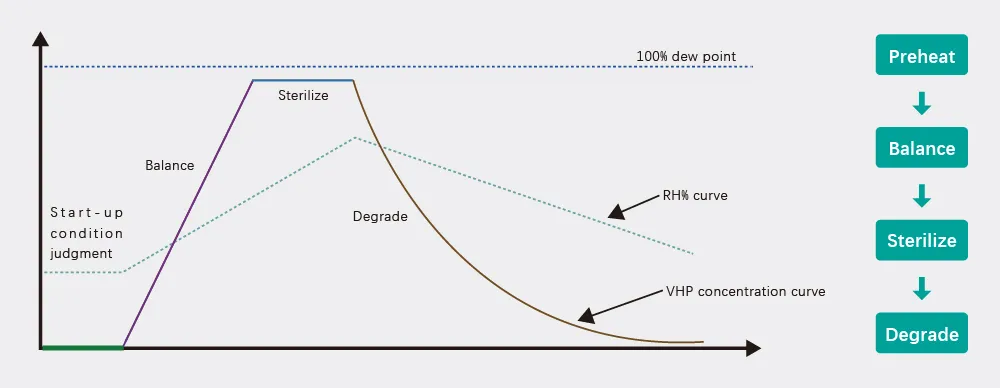
VHP is a technology that vaporizes the liquid hydrogen peroxide into hydrogen peroxide vapor and uses the vaporized hydrogen peroxide (VHP) to sterilize the surface of the object at low temperature. It has broad-spectrum bactericidal properties and can effectively kill all types of microorganisms such as bacteria, fungi, molds, viruses, and bacterial spores.

Using the process control method of D-value for sterilized microorganism. Generally set to 6LOG reduction program.
Generally set to 12LOG overkill program for maximum sterilization security.
Controlled by specific concentration and time conditions obtained from parameter development.
Achieved through HEPA filter filtration to realize transfer without full sterilization, partially replacing standard pass box functions.

| Model | Inner Cavity Dimensions | External Dimensions | Power (kW) | Weight (kg) |
|---|---|---|---|---|
| HPB-216 | 600*600*600mm | 1050*650*1800mm | 2.0 | 280 |
| HPB-288 | 600*600*800mm | 1050*650*2000mm | 2.2 | 300 |
| HPB-512 | 800*800*800mm | 1250*850*2000mm | 2.3 | 350 |
| HPB-960 | 800*1000*1200mm | 1250*1050*2200mm | 2.8 | 400 |
| HPB-2688 | 1200*1400*1600mm | 1850*1450*2150mm | 4.0 | 600 |
Note: Custom configurations are available upon request for non-standard requirements.
Utilizing advanced CNC equipment, laser cutting, and fully automatic welding systems. Our facilities include filtration, sheet metal, welding, and assembly workshops.








VHP (Vaporized Hydrogen Peroxide) sterilization works by vaporizing liquid hydrogen peroxide and using the gas to eliminate microorganisms on surfaces at low temperatures, eventually breaking down into water and oxygen.
No. After the sterilization cycle, the VHP quickly degrades into H2O (water) and O2 (oxygen), making it non-toxic and environmentally friendly.
LOGA typically refers to a 6LOG reduction program, while LOGB is a 12LOG overkill program designed for environments requiring the highest level of sterilization assurance.
Yes, we provide non-standard customized products. You can specify inner cavity and external dimensions to suit your cleanroom layout.
It is widely used in biopharmaceuticals, life sciences, healthcare, laboratory research, and food safety industries where sterile transfer is critical.
Our VHP equipment is manufactured under ISO9001-2015 standards and holds international certifications including CE and UL, meeting GMP and FDA requirements.