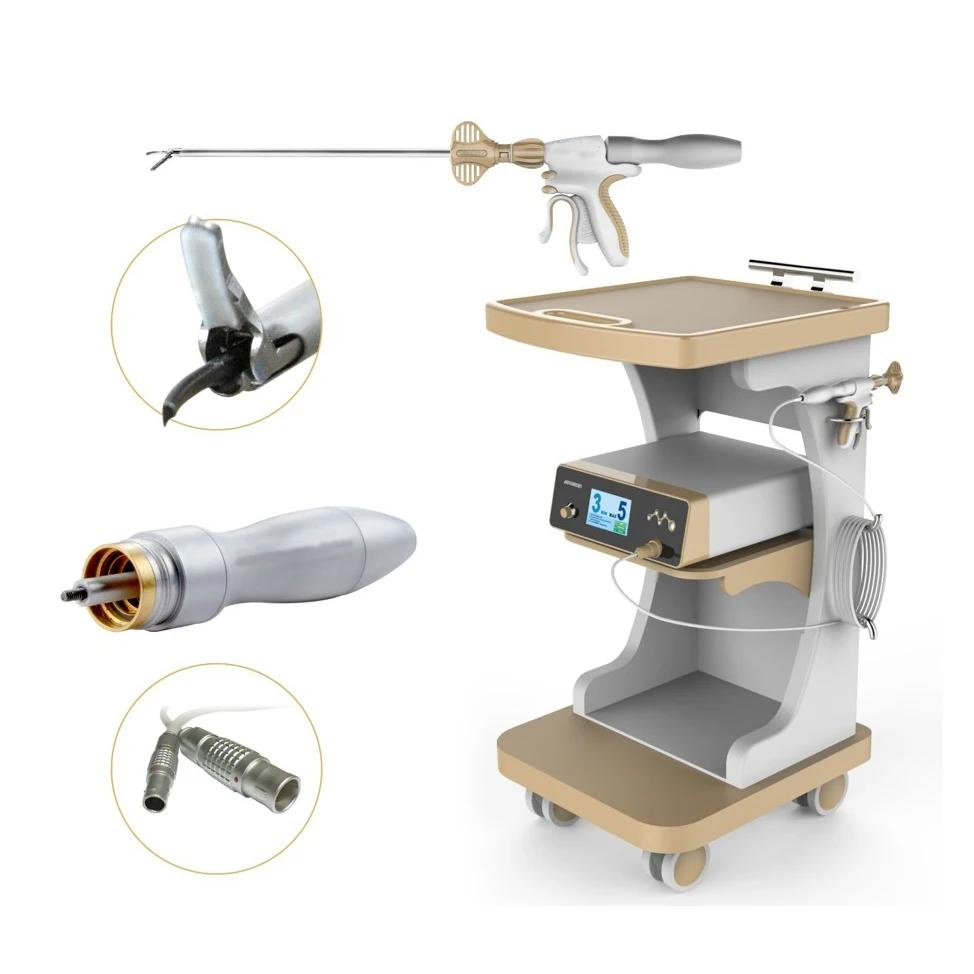




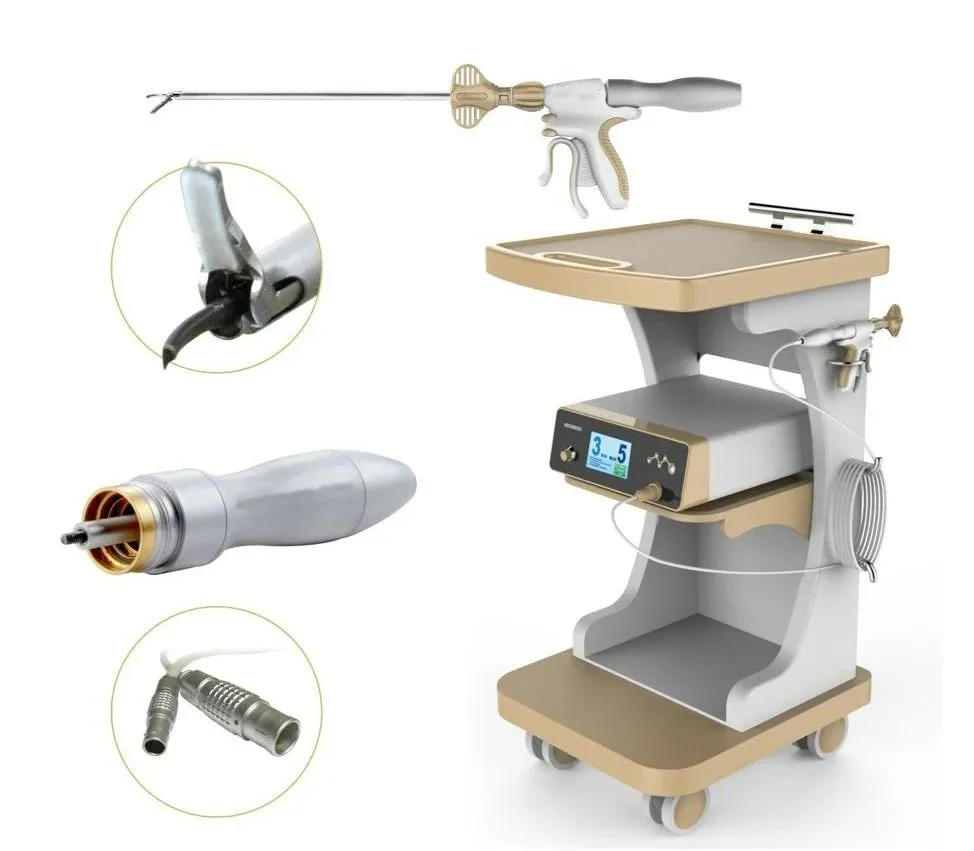
1 / 5










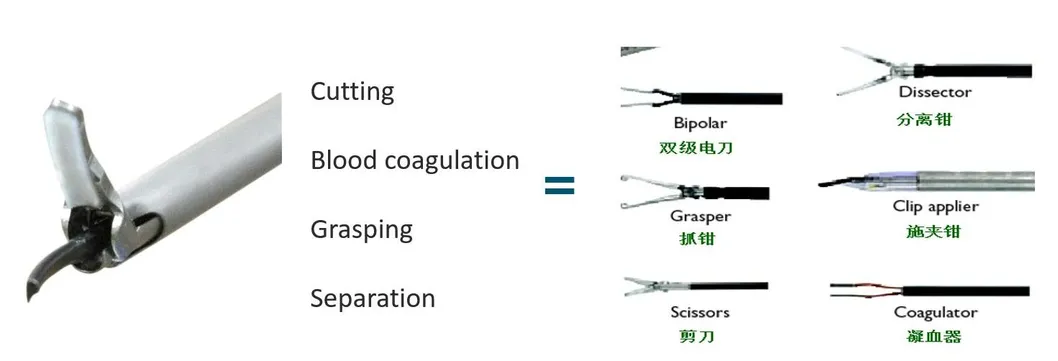


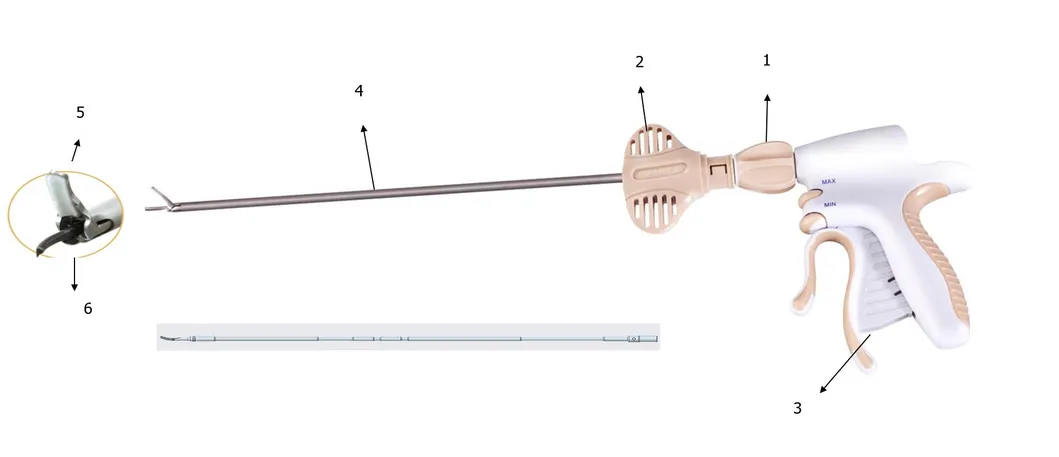
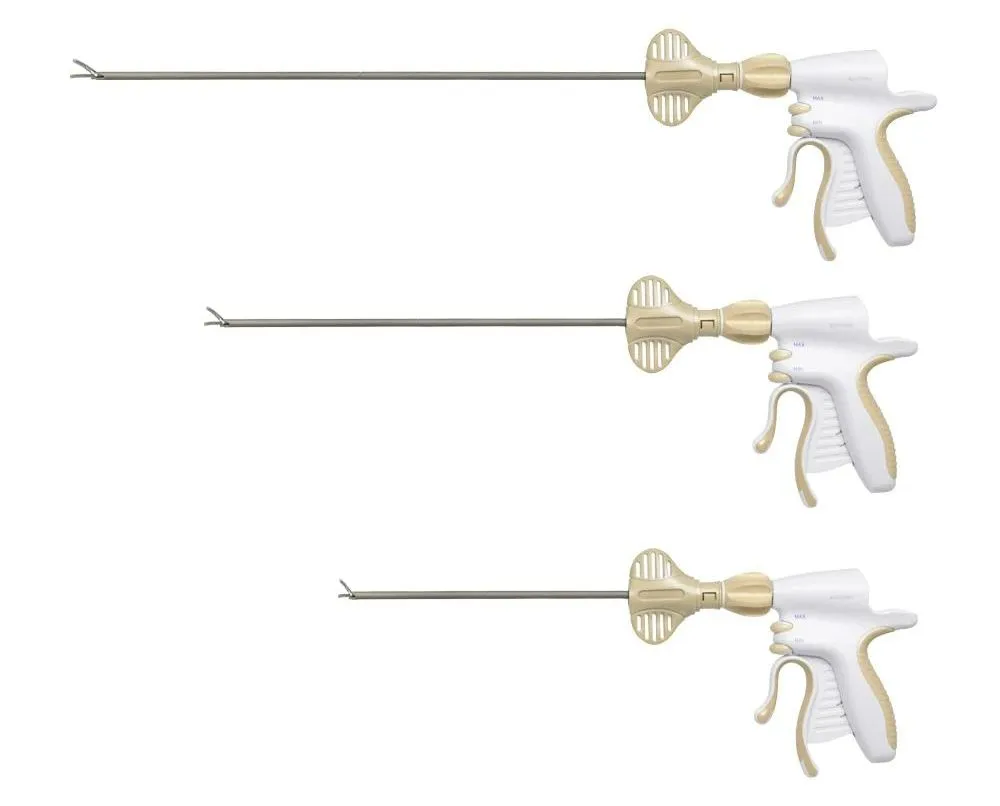
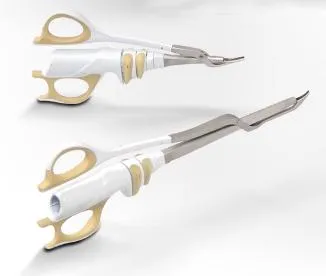
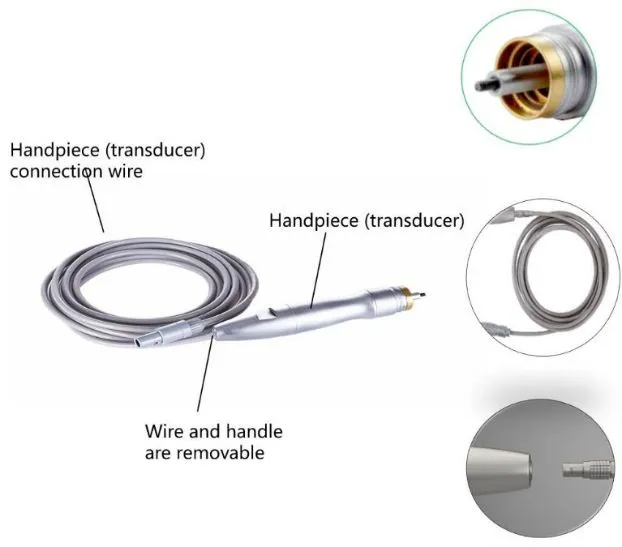


1. Global Regulatory Expertise: Our leadership team possesses over 20 years of experience in medical device quality management and international registrations, including successful applications for FDA 510K and CE marks.
2. Specialized Teams: We maintain professional R&D, quality control, and sales teams that stay updated with the latest global medical industry technologies.
3. Partnership Opportunities: We welcome global distributors and agents, supporting them with competitive pricing and high-quality products for local market development.
4. Production Support: Beyond finished devices, we can supply parts and automatic production machinery to help partners establish their own local manufacturing facilities.