France is a pioneer in the field of Minimally Invasive Surgery (MIS). Home to the world-renowned IRCAD (Institut de Recherche contre les Cancers de l'Appareil Digestif) in Strasbourg, the French medical ecosystem serves as a global incubator for endoscopic innovation. The demand for high-quality endoscopic instruments in French hospitals—both public (AP-HP) and private clinics—is driven by a rigorous commitment to patient safety and technological advancement.
In France, the supply of endoscopic instruments is governed by the ANSM (National Agency for the Safety of Medicines and Health Products). Suppliers and factories must adhere to the stringent EU Medical Device Regulation (MDR 2017/745). This ensures that every laparoscope, forceps, and scalpel used in French operating theaters meets the highest levels of biocompatibility and mechanical precision.
French surgical centers in Paris, Lyon, and Marseille are increasingly integrating robotic platforms with traditional endoscopic instruments. This hybrid approach has created a niche for "robotic-compatible" manual instruments, where precision-engineered tips and ergonomic handles are paramount for secondary port interventions.
There is a significant shift in France towards "Green Hospitals." While disposable instruments offer sterility convenience, many French procurement departments are returning to high-quality reusable endoscopic instruments that can withstand hundreds of autoclave cycles, reducing medical waste and long-term costs.
French urologists and gastroenterologists are demanding instruments that complement 4K imaging systems. This means instruments must have anti-reflective coatings and ultra-slim profiles to allow for maximum visibility during complex procedures like Ureteropyeloscopy (URS).
The trend towards "scarless" surgery is pushing French factories to produce instruments with diameters as small as 2mm to 3mm, used extensively in pediatric endoscopy and advanced laparoscopy.
Dongguan LeienCare Tech Co., Ltd. is a professional manufacturer specializing in surgical instruments, disposable surgical supplies, and sterile medical products, serving global healthcare markets as a reliable OEM supplier. The company is committed to delivering high-quality, safe, and efficient medical solutions for hospitals, clinics, laboratories, and healthcare distributors worldwide.
LeienCare Tech offers a comprehensive product portfolio that includes a wide range of surgical instruments, operating room consumables, sterile packaging solutions, and disposable medical supplies designed to meet diverse clinical requirements. All products are manufactured under strict quality control systems and comply with international medical standards to ensure precision, reliability, and patient safety in critical medical environments.



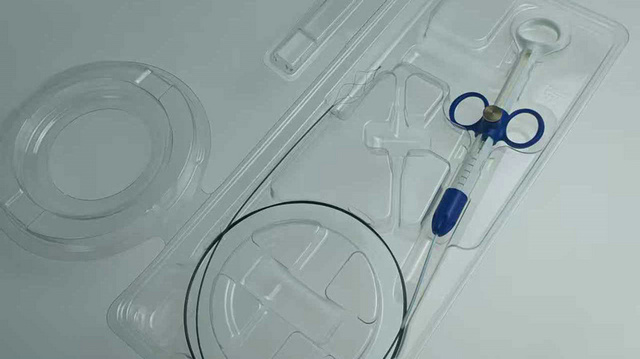
With advanced production facilities, precision manufacturing capabilities, and a strong focus on hygiene and sterilization processes, Dongguan LeienCare Tech Co., Ltd. maintains consistent product quality and scalable production capacity. The company also provides flexible OEM and ODM services, supporting customization in product design, materials, packaging, and private labeling to meet specific customer and market needs.
Driven by innovation, quality assurance, and customer satisfaction, LeienCare Tech continues to expand its global reach. The company is dedicated to supporting healthcare professionals with dependable surgical and disposable medical products that enhance operational efficiency and contribute to improved patient care outcomes.




In French University Hospitals (CHU), there is a heavy reliance on specialized instruments for training future surgeons. Precision-engineered graspers and dissectors are essential for simulation-based learning and live surgical interventions in oncology and bariatric surgery.
Private clinics in regions like the French Riviera or Ile-de-France prioritize high-turnover efficiency. Disposable staplers and catheters (with CE Mark and ISO 13485) are preferred here to minimize sterilization bottlenecks and ensure rapid patient discharge.

Establishing a foothold in the French medical market requires more than just high-quality products; it requires Authority and Reliability. At LeienCare, we understand that French suppliers need partners who can navigate the complexities of CE marking and localized medical documentation. Our manufacturing processes are designed to mirror the excellence expected by the Société Française de Chirurgie Endoscopique (SFCE).
Experience in the field of endoscopy is measured by the tactile feedback of the instrument. Our Clip Applicators and Maryland Forceps are tested for ergonomic comfort, ensuring that during long procedures in French ORs, surgeon fatigue is minimized. This focus on "Expertise" (the E in E-E-A-T) is what differentiates a standard factory from a global surgical partner.
Furthermore, our Ureteropyeloscopy (URS) instruments are engineered with flexible fibers and high-definition optics to support the growing trend of lithotripsy and oncology treatments in the French urology sector. By providing both reusable and disposable options, we empower French hospitals to balance their budgets while maintaining the highest standards of sterile surgical care.
Contact our French export specialists today for customized OEM solutions and bulk pricing.
Send Inquiry Now