The global surgical suture materials market is currently experiencing a transformative phase, driven by an aging global population and the increasing volume of surgical procedures worldwide. As of 2024, the industry is valued at over USD 4.5 billion, with a steady compound annual growth rate (CAGR). This growth is fueled not only by traditional open surgeries but also by the rising demand for specialized sutures used in minimally invasive and robotic-assisted procedures.
Industrial stakeholders are shifting focus toward biocompatible and bioabsorbable polymers. The demand for high-tensile strength, reduced tissue reaction, and predictable absorption rates has made materials like Polyglycolic Acid (PGA) and Polylactic-co-glycolic Acid (PGLA) the industry standards. Meanwhile, non-absorbable materials like Silk, Nylon, and Polypropylene remain critical for cardiovascular and orthopedic applications where long-term structural integrity is paramount.
Geographically, while North America and Europe maintain high consumption rates due to advanced healthcare infrastructures, the Asia-Pacific region—led by China—has emerged as the primary manufacturing hub. Chinese factories have evolved from low-cost providers to high-tech innovators, integrating AI-driven quality control and precision extrusion technologies to meet stringent international standards such as CE and ISO 13485.
The commercial landscape is also seeing a consolidation of supply chains. Global medical distributors are increasingly seeking direct partnerships with manufacturers to mitigate logistics risks and ensure a steady supply of sterile surgical consumables, especially in the post-pandemic era where healthcare resilience is a top priority.
One of the most significant trends is the development of sutures coated with antimicrobial agents (like Triclosan) to prevent Surgical Site Infections (SSIs). Future iterations are exploring drug-eluting sutures that release localized anesthesia or growth factors to accelerate the healing process.
Integration of electronic sensors into suture materials is on the horizon. These "Smart Sutures" can monitor wound tension, temperature, and pH levels, wirelessly transmitting data to healthcare providers to detect early signs of infection or dehiscence.
As hospitals aim for "Green OR" initiatives, manufacturers are researching eco-friendly packaging and biodegradable materials that reduce the environmental footprint of disposable medical supplies without compromising sterility or safety.
The rise of knotless wound closure using barbed sutures has revolutionized laparoscopic surgery. These materials allow for more even tension distribution and faster closure times, which is essential for reducing anesthesia duration for patients.
Global Procurement Requirements: Modern medical buyers—ranging from large hospital groups to specialized distributors—prioritize three pillars: Regulatory Compliance, Supply Chain Transparency, and Customization. In the EU, compliance with the new Medical Device Regulation (MDR) is non-negotiable. In emerging markets, the focus is often on balancing high-quality clinical outcomes with cost-efficiency.
Localized Application Scenarios:
Dongguan LeienCare Tech Co., Ltd. is a professional manufacturer specializing in surgical instruments, disposable surgical supplies, and sterile medical products, serving global healthcare markets as a reliable OEM supplier. The company is committed to delivering high-quality, safe, and efficient medical solutions for hospitals, clinics, laboratories, and healthcare distributors worldwide.
LeienCare Tech offers a comprehensive product portfolio that includes a wide range of surgical instruments, operating room consumables, sterile packaging solutions, and disposable medical supplies designed to meet diverse clinical requirements. All products are manufactured under strict quality control systems and comply with international medical standards to ensure precision, reliability, and patient safety in critical medical environments.
With advanced production facilities, precision manufacturing capabilities, and a strong focus on hygiene and sterilization processes, Dongguan LeienCare Tech Co., Ltd. maintains consistent product quality and scalable production capacity. The company also provides flexible OEM and ODM services, supporting customization in product design, materials, packaging, and private labeling to meet specific customer and market needs.
Driven by innovation, quality assurance, and customer satisfaction, LeienCare Tech continues to expand its global reach. The company is dedicated to supporting healthcare professionals with dependable surgical and disposable medical products that enhance operational efficiency and contribute to improved patient care outcomes.



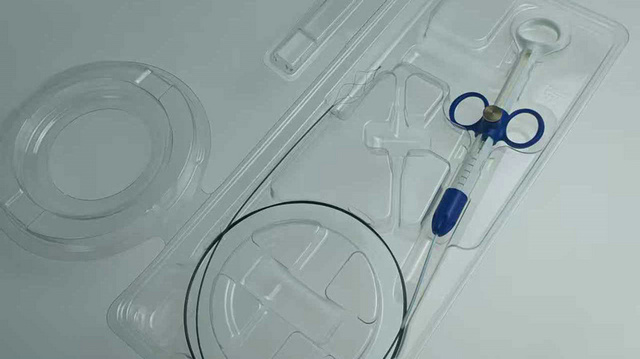




At the heart of every surgical procedure is the material that holds the tissue together. As a leading China Suture Factory, we delve deep into polymer science. Absorbable sutures are engineered to lose their tensile strength at a rate that matches the tissue's healing curve. For instance, our PGA (Polyglycolic Acid) sutures are synthesized through the polymerization of glycolide, offering high initial tensile strength and a predictable absorption period of 60 to 90 days via hydrolysis.
Non-absorbable sutures, such as our medical-grade Silk and Nylon, are treated with specialized coatings to reduce capillarity. Silk sutures are degummed and coated with wax or silicone to ensure smooth passage through delicate tissues, making them ideal for ophthalmic and neurological surgeries where precision is paramount.
A suture is only as good as the needle it is attached to. Our manufacturing process involves the use of high-quality 300 and 400 series stainless steel. We employ advanced grinding techniques to create various needle points: Taper Point, Blunt Point, Triangular Cutting, and Reverse Cutting. Each needle is rigorously tested for "ductility" and "sharpness." A needle must be sharp enough to penetrate tissue with minimal force but ductile enough to bend without breaking under extreme stress.
The attachment between the thread and the needle (the "swage") is a critical point of failure. We utilize laser-drilled swaging technology to ensure a seamless transition, which minimizes tissue trauma during the suturing process—a key requirement for minimally invasive surgeries.
Sterility is the cornerstone of patient safety. Our facility utilizes Ethylene Oxide (EO) sterilization and Gamma Radiation, depending on the material's sensitivity. Every batch undergoes biological indicator testing to confirm 100% sterility before dispatch. Our cleanrooms are maintained at Class 100,000 (ISO 8) standards, ensuring that environmental contaminants are strictly controlled during the assembly and packaging phases.
Furthermore, our quality control team performs "Pull Tests" to measure the breaking strength of the suture and the attachment strength of the needle. We adhere to USP (United States Pharmacopeia) and EP (European Pharmacopoeia) standards, ensuring that our products are globally interchangeable with premium brands.
As an OEM partner, Dongguan LeienCare Tech Co., Ltd. empowers global brands to expand their product lines without the capital expenditure of building factories. We provide "Private Label" solutions where we handle everything from material sourcing and sterilization to custom packaging design. This allows our clients to focus on clinical training and market distribution while we ensure the technical reliability of the products.
Our R&D department works closely with surgeons to develop custom suture-needle combinations for niche applications, such as cardiovascular valve replacement or pediatric reconstructive surgery. This collaborative approach ensures that our products are not just "commodities" but clinical tools that save lives.